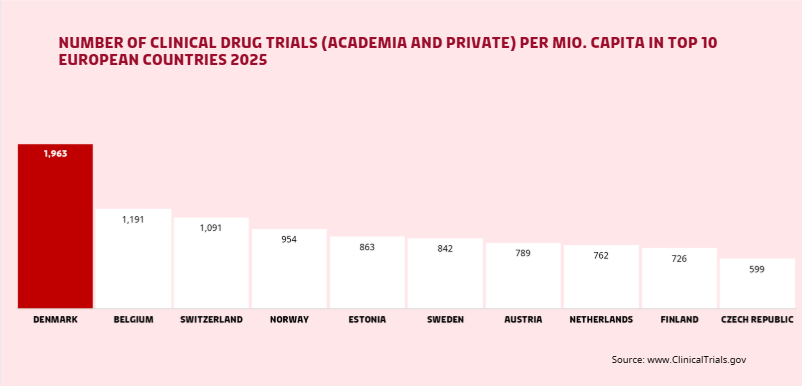
With the Strategy for Life Science towards 2030 (New Danish life science strategy), published in November 2024, the Danish government committed to establishing a fourth Medical Research Ethics Committee. The goal? To ensure Phase 1 trials are processed within just 14 days, reinforcing Denmark’s position as one of the most attractive locations in Europe to conducting clinical trials.
On 14. august 2025, the new 14-day assessment process for phase I trials was officially launched. It includes concurrent regulatory and ethics assessments of applications, a collaboration between the Danish Medicines Agency (DMA) and the Danish Medical Research Ethics Committees (MREC).
Initially, the fast-track timeline will automatically apply to all mono-national phase I and phase I-II trials. For multinational trials, accelerated approval will depend on other Member States matching Denmark’s speed. The DMA and MREC encourage other countries to adapt similar rapid assessment models, improving Europe’s competitiveness as a whole.